The iAssay® System is designed for use for Point of Care Testing (POCT) by healthcare practitioners wherever the patient is located: at the hospital, doctor’s office, pharmacy, and at a patient’s home. iAssay®will begin with patient home visits, then move into other markets.
One device (instead of a closet full of single function readers) provides a patient test panel in minutes next to the patient, eliminating the time and cost of transporting samples to the nearest hospital for processing in hours or days.
Faster test results mean that patients can be treated now instead of later, improving patient outcomes.
Drug Screening with the CyberReader ® Model 2030
Multi Format Demonstration with Early Prototype
Test, patient, and local wearable data is aggregated by the iAssay® System and HIPAA compliantly pushed as a single Datastream into The Cloud enabling auto-EMR population, auto-reimbursement billings, quality control, and remote physician monitoring.

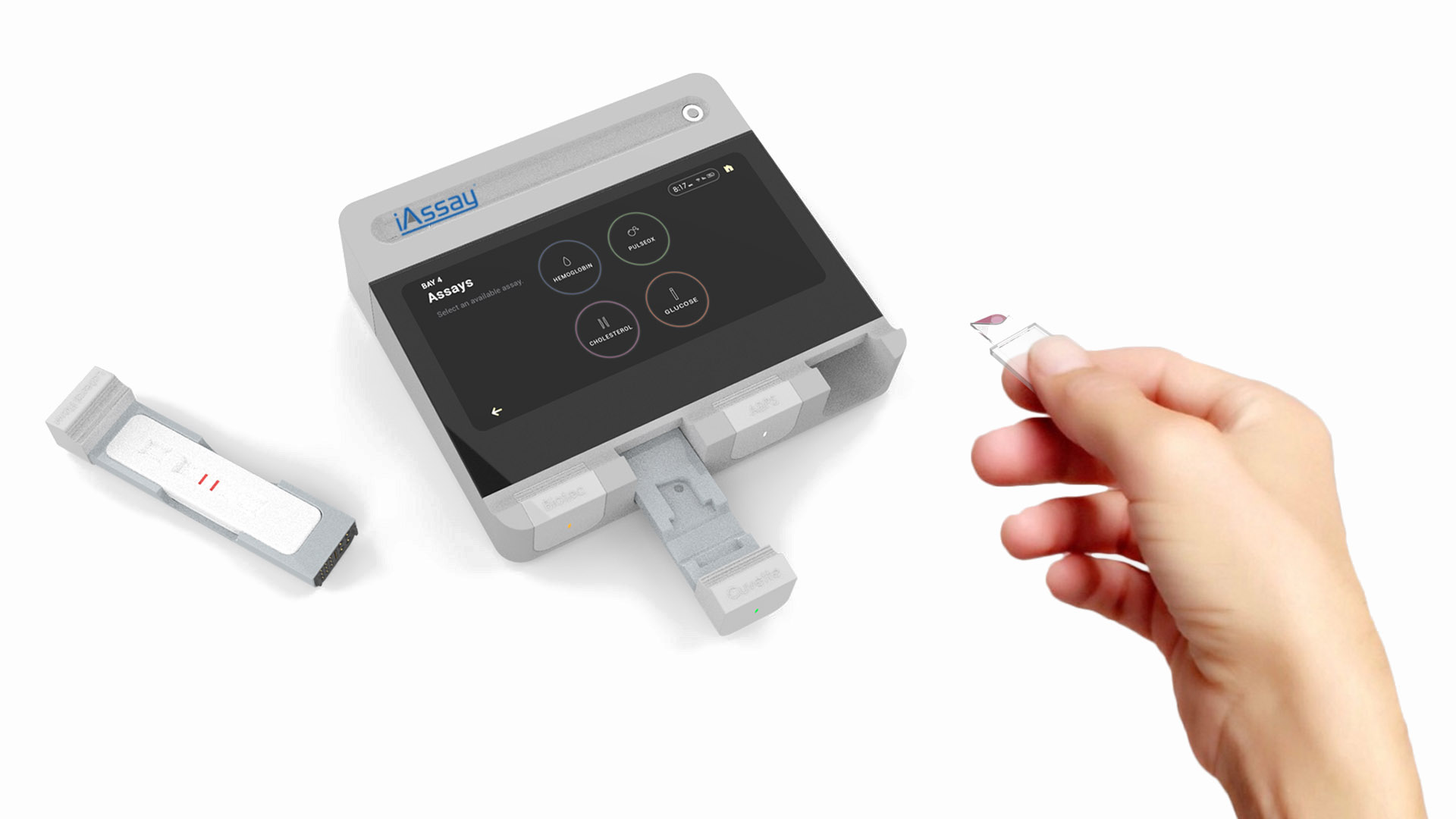
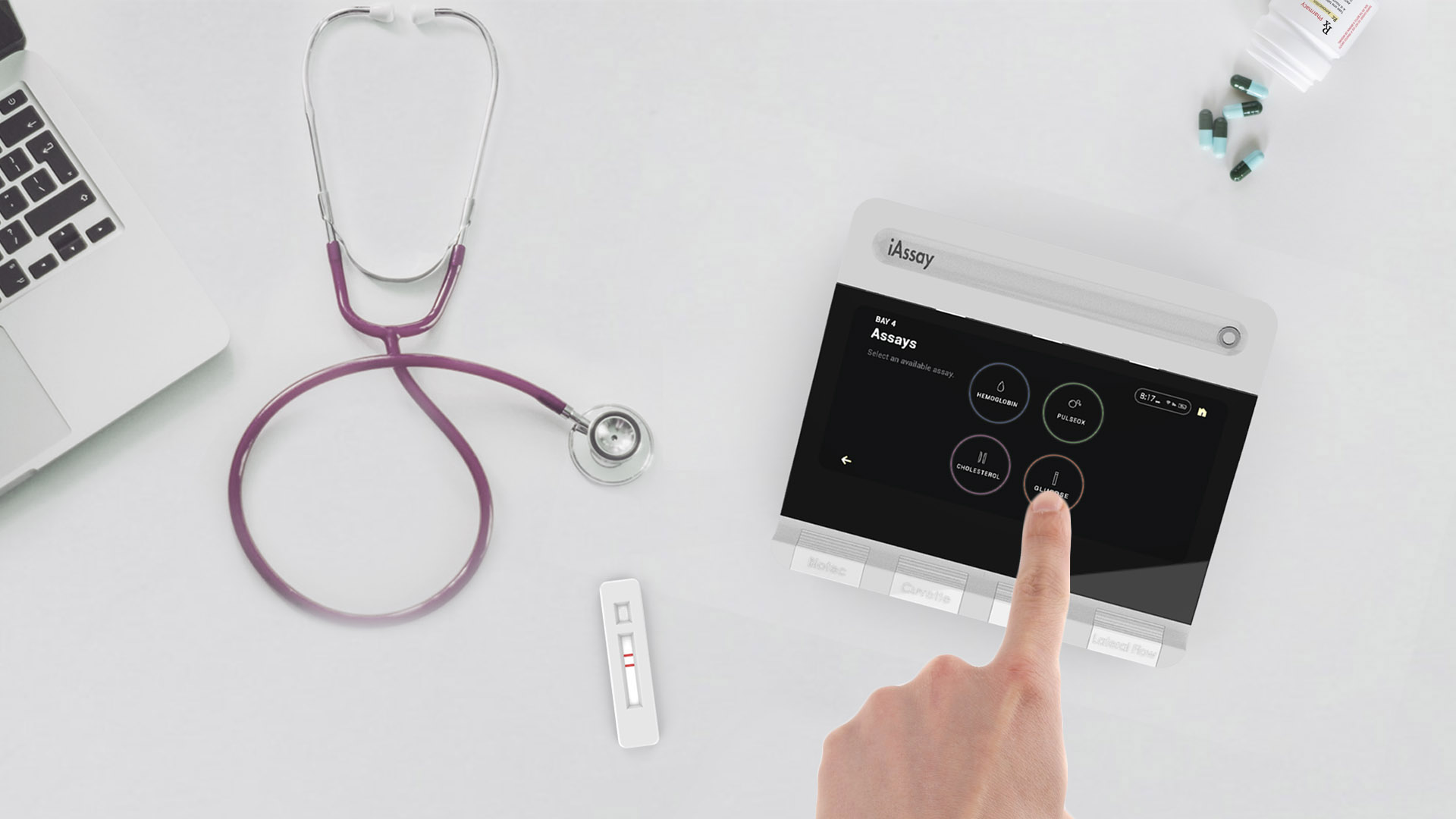
iAssay® supports popular assay (test) cartridge formats adapted in collaboration with assay cartridge partners by simply plugging the appropriate “Adaptor Module” into the Open Platform designed and produced by iAssay®. Assay cartridges are then inserted into the Adaptor Module.
The combination of platform and adaptor operates the test cartridge exactly as the assay cartridge developer/manufacturer intends, but at a small fraction of the price of a single function reader. iAssay® will initially provide POC testing for diabetes (A1C), electrolytes, cardiac markers, hematology, and PT/INR.
In addition to Adaptor Modules that adapt POCT test cartridges to the platform, Functional Modules (e.g. high-speed microprocessor, satellite communications, imaging, and fluid control) allow for iAssay® System expansion by just inserting a plug-in.
The capabilities of the iAssay® System are essentially limitless from a Point of Care standpoint because of full system programmability and plug-ins. All Point of care assays can now be run on one inexpensive platform, eliminating the need for multiple readers.
 |
 |
